Several U.S. senators are questioning the Food and Drug Administration’s work with a consulting firm that helped businesses sell prescription painkillers during the nation's overdose crisis.
Democrat Maggie Hassan of New Hampshire and Republican Chuck Grassley of Iowa, among others, have asked the FDA about potential conflicts of interest in its work with McKinsey and Co.
The senators sent a letter Monday to the FDA noting that the agency paid McKinsey more than $140 million after hiring it several times starting in 2008.
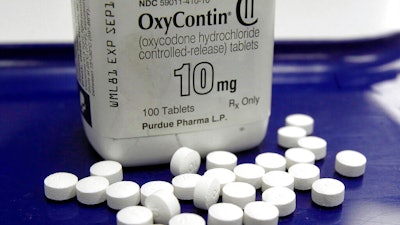
While helping the government, McKinsey “also worked for a wide range of actors in the opioid industry, including many of the companies that played a pivotal role in fueling the opioid epidemic that our country now faces,” the letter said.
An FDA representative said the agency has received the letter and would respond directly to the senators.
Earlier this year, McKinsey agreed to almost $600 million in settlements with nearly every state and five U.S. territories over its work with drugmakers on prescription opioid sales.
Opioids, which also include illegal substances like heroin, have been linked to almost a half million deaths in the U.S. since 2000, a problem that has deepened during the COVID-19 pandemic.
Under its contracts with the FDA, McKinsey worked with the Center for Drug Evaluation and Research, the agency's main division for approving prescription opioids and other drugs, the letter states.
While working with the FDA, McKinsey also advised opioid manufacturers on avoiding oversight from the regulator, according to the letter. The consultant advised Purdue Pharma on how to soften an FDA drug safety program that required companies to communicate risks to patients, care providers and pharmacists.
The senators noted that the final program announced in 2012, “was largely devoid of the restrictions the FDA had initially proposed.”
The senators asked the FDA to answer several questions and provide documents by Sept. 20. They want to know how the agency checks for conflicts of interest and what its current relationship is with McKinsey.
They also asked what disclosures McKinsey made to the agency about potential conflicts and when the FDA became aware that McKinsey’s clients included “several major opioid distributors and retailers.”
Four Democratic senators joined Hassan and Grassley in signing the letter. They include West Virginia’s Joe Manchin, Rhode Island’s Sheldon Whitehouse and Massachusetts Sens. Elizabeth Warren and Edward Markey.