There is dynamic growth in the use of low-power IIoT-connected remote wireless devices. These applications include asset tracking, supervisory control and data acquisition, environmental monitoring, AI, and M2M, to name a few.
Battery-powered solutions are expanding opportunities for bringing cost effective power to hard-to-access locations and extreme environments. This is especially important where hard-wired solutions are cost prohibitive, making the higher initial cost of an industrial grade battery cost justified based on the ability to achieve higher reliability and a lower cost of ownership.
Meeting the demands of low-power devices
There are two main types of low-power devices: those that draw average energy including background current and pulses measurable in microamps, which are best suited for an industrial grade primary (non-rechargeable) lithium battery; and those that draw average energy including background current and pulses measurable in milliamps, which would prematurely exhaust a primary battery. These niche applications are typically better served by some form of energy harvesting device in combination with an industrial grade rechargeable Lithium-ion (Li-ion) battery to store the harvested energy.
Specifying a battery invariably involves trade-offs involving numerous criteria, including: the amount of current consumed during active mode (including the size, duration, and frequency of pulses); energy consumed during stand-by mode (the base current); storage time (as normal self-discharge during storage diminishes capacity); thermal environments (including storage and in-field operation); and equipment cut-off voltage, which drops as cell capacity is exhausted or during prolonged exposure to extreme temperatures. Often, a critical consideration is the annual self-discharge rate of the cell, which often exceeds the energy required to operate the device.
As the lightest non-gaseous metal, with a high intrinsic negative potential that exceeds all others, lithium offers the highest specific energy (energy per unit weight) and energy density (energy per unit volume) of all commercially available chemistries. Lithium cells operate within a normal operating current voltage range of 2.7 to 3.6V. This chemistry is non-aqueous, and thus less likely to freeze in extremely cold temperatures.
There are numerous choices among primary (non-rechargeable) chemistries, including iron disulfate (LiFeS2), lithium manganese dioxide (LiMNO2), lithium thionyl chloride (LiSOCl2), alkaline, and lithium metal oxide chemistry (Table 1).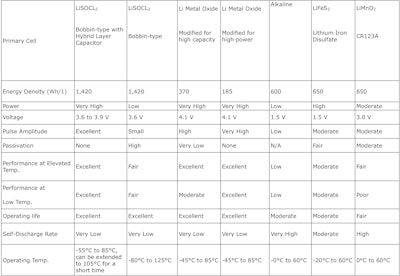 Table 1.
Table 1.
Why battery self-discharge is so crucial
Self-discharge is common to all batteries, as chemical reactions sap energy even when the cell is unused or in storage. Self-discharge can be sizably reduced by harnessing the passivation effect.
Passivation occurs only in LiSOCl2 batteries, where a thin film of lithium chloride (LiCl) forms on the surface of the anode, separating it from the electrode, thereby limiting the chemical reactions that cause self-discharge. When a load is placed on the cell, the passivation layer causes high initial resistance and a temporary dip in voltage until the discharge reaction begins to dissipate the passivation layer: a process that repeats each time the load is removed.
Numerous factors can impact the passivation effect, including: current discharge capacity; the length of storage and storage temperature; discharge temperature; and prior discharge conditions, as partially discharging a cell and then removing the load increases the amount of passivation relative to when the cell was new. Passivation is beneficial to extending battery life, but too much of it can restrict energy flow.
Self-discharge is also impacted by the cell’s current discharge potential, how the cell is manufactured, and the quality and purity of the raw materials. For instance, a higher quality bobbin-type lithium thionyl chloride (LiSOCl2) cell can feature a self-discharge rate as low as 0.7% per year, retaining 70% of its original capacity after 40 years. By contrast, a lower quality bobbin-type LiSOCl2 cell can experience a self-discharge rate of up to 3% per year, loosing 30% of its initial capacity every 10 years, making 40-year battery life impossible.
Lithium thionyl chloride (LiSOCl2) chemistry can be constructed in two ways: bobbin-type or spiral wound. Spiral wound cells deliver faster energy flow with the trade off of a higher self-discharge rate. Bobbin-type LiSOCl2 batteries are particularly well suited for certain low-power applications that can benefit from its higher capacity and energy density, wider temperature range (-80°C to 125° C), and exceptionally low self-discharge.
Two-way wireless communications requires high pulses
Battery-powered devices increasingly require periodic high pulses to power two-way wireless communications. To conserve energy, these devices must be intelligently designed using a low power communications protocol (e.g., WirelessHART, ZigBee, or LoRa), a low-power chipset, and proprietary energy-saving techniques.
Due to their low rate design, standard bobbin-type LiSOCl2 cells cannot deliver the high pulses required to power two-way wireless communications. This challenge can be overcome with the addition of a patented hybrid layer capacitor (HLC). The bobbin-type LiSOCl2 cell delivers background current during standby mode while the HLC works like a rechargeable battery to generate high pulses up to 15A. The patented HLC also features a unique end-of-life voltage plateau that allows for “low battery” status alerts (Figure 2). Figure 2: Bobbin-type liSOCl2 batteries can be combined with a patented hybrid layer capacitor (HLC) to offer up to 40-year operating life while also providing high pulses to power two-way wireless communications.
Figure 2: Bobbin-type liSOCl2 batteries can be combined with a patented hybrid layer capacitor (HLC) to offer up to 40-year operating life while also providing high pulses to power two-way wireless communications.
Supercapacitors often perform this function in consumer electronics but are rarely found in industrial applications due to inherent drawbacks such as short-duration power; linear discharge qualities that do not permit full discharge of available energy; low capacity; low energy density; and a very high self-discharge rate of up to 60% per year. Supercapacitors linked in series also require cell-balancing circuits that are bulky, expensive, and extract additional energy to further shorten their operating life.
Here are some real-life examples involving bobbin-type LiSOCl2 batteries:
Cryoegg: Researchers studying the impact of climate change and rising sea levels on deep water channels beneath glaciers in Greenland and Antarctica rely upon Cryoegg, an underwater sensor, to monitor temperature, pressure, and electrical connectivity (Figure 3). Cryoegg eliminates the need for hard-wired cables that are bulky, expensive, and easily damaged by glacial movement. Bobbin-type LiSOCl2 cells were chosen for their high capacity, high energy density, wide temperature range, and high pulse capabilities. Figure 3: Researchers from Cardiff University studying water channels beneath glaciers developed the Cryoegg, which monitors temperature, pressure, and electrical connectivity, transmitting data underwater via radio waves.Mauro Werder/Cardiff University
Figure 3: Researchers from Cardiff University studying water channels beneath glaciers developed the Cryoegg, which monitors temperature, pressure, and electrical connectivity, transmitting data underwater via radio waves.Mauro Werder/Cardiff University
The Cryoegg wireless sensor utilizes 169 MHz Wireless M-Bus radio waves that were popularized for use in AMR/AMI water and gas utility meter transmitter units (MTUs) to reduce the cost of ownership. By offering extended battery life, this chemistry helps prevent wide-scale battery failures that are highly disruptive to billing systems and remote start-up/shut-off capabilities.
Oceantronics: Transporting scientific equipment across the Arctic can be challenging. For this reason, Oceantronics redesigned the battery pack for its GPS/ice buoy by eliminating the need for an oversized battery pack consisting of 380 alkaline D cells with a far more compact, lightweight, and cost-efficient solution using 32 bobbin-type LiSOCl2 cells and 4 HLCs. Oceantronics achieved a 90% reduction in size and weight (54 kg down to 3.2 kg), making their GPS/ice buoys more easily transportable by helicopter (Figures 4 and 5). Converting from alkaline to LiSOCl2 chemistry also increased operating life many fold.  Figure 5: Oceantronics redesigned the battery pack for its GPS/ice buoy, replacing 380 alkaline D cells with a far smaller, lighter, and more economical battery pack using 32 bobbin-type LiSOCl2 cells and four HLCs. Size and weight were reduced by 90% (54 kg down to 3.2 kg), making the GPS/ ice bouy far easier to transport via helicopter to icebergs near the North Pole.Sigrid Salo NOAA/PMEL; Inset (Figure 4): Oceantronics
Figure 5: Oceantronics redesigned the battery pack for its GPS/ice buoy, replacing 380 alkaline D cells with a far smaller, lighter, and more economical battery pack using 32 bobbin-type LiSOCl2 cells and four HLCs. Size and weight were reduced by 90% (54 kg down to 3.2 kg), making the GPS/ ice bouy far easier to transport via helicopter to icebergs near the North Pole.Sigrid Salo NOAA/PMEL; Inset (Figure 4): Oceantronics
Southwire: Reducing size and weight is important to utility line crews installing line/connector sensors that monitor temperature, catenary, and line current, providing an immediate warning if a transmission line goes down (Figure 6). Bobbin-type LiSOCl2 cells enables a more portable and lightweight (3.5 lbs.) solution that is capable of handling extreme temperatures (-40° C to 50° C). Southwire line/connector sensors provide months of back-up power if no line current is detected. Figure 6: Bobbin-type LiSOCl2 cells reduced the size and weight of Southwire’s line/connector sensors that monitor the status of electric power transmission lines. These batteries enable the sensors to be compact and lightweight (3.5 lb.), more easily portability by line crews climbing tall towers.Southwire
Figure 6: Bobbin-type LiSOCl2 cells reduced the size and weight of Southwire’s line/connector sensors that monitor the status of electric power transmission lines. These batteries enable the sensors to be compact and lightweight (3.5 lb.), more easily portability by line crews climbing tall towers.Southwire
Sometimes, energy harvesting is required
Applications that draw current measurable in milli-amps can quickly exhaust a primary lithium battery, requiring the use of an energy harvesting device in tandem with an industrial grade rechargeable Lithium-ion (Li-ion) battery.
One example is Cattlewatch, which combines small solar (PV) panels and Li-ion batteries to create mesh networks that track the location, health, and safety of animal herds (Figure 7).  Figure 7: Cattlewatch collar tag transmitters combine a small PV panel with an industrial grade rechargeable Li-ion battery, creating a lightweight solution that transits data about health status and location remotely to ranchers.Cattlewatch
Figure 7: Cattlewatch collar tag transmitters combine a small PV panel with an industrial grade rechargeable Li-ion battery, creating a lightweight solution that transits data about health status and location remotely to ranchers.Cattlewatch
Solar/Li-ion hybrids also power smart parking meter fee collection systems that are equipped with AI-enabled sensors to identify open parking spots (Figure 8). Figure 8: IPS solar-powered parking meters utilize industrial grade rechargeable Li-ion batteries to operate for up to up to 20 years while providing 24/7/365 system reliability and connectivity for smart parking meters.IPS Group
Figure 8: IPS solar-powered parking meters utilize industrial grade rechargeable Li-ion batteries to operate for up to up to 20 years while providing 24/7/365 system reliability and connectivity for smart parking meters.IPS Group
Inexpensive consumer-grade rechargeable Li-ion cells are relatively short lived (5 years and 500 recharge cycles), with a limited temperature range (0-40° C), and cannot deliver high pulses for two-way wireless communications. By contrast, industrial grade Li-ion batteries can operate up to 20 years and 5,000 full recharge cycles, offering an expanded temperature range (-40° to 85° C), and able to deliver high pulses (Table 9).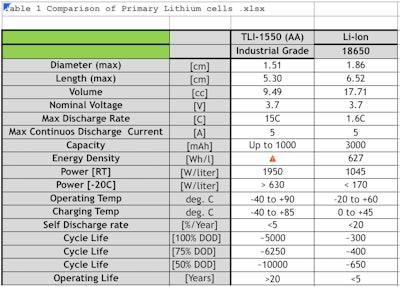 Table 9.
Table 9.
If your battery needs to last as long as the device, then you need to carefully choose the chemistry that best matches application requirements. You should perform thorough due diligence when evaluating potential battery suppliers, requesting well-documented long-term test results, actual in-field performance data under similar environmental conditions, and numerous customer references.
Specifying a more rugged long-life battery pays off in the long-term by increasing product reliability and reducing your total cost of ownership.
Sol Jacobs is the vice president and general manager of Tadiran Batteries.